About Us
BBMRI.sk – The Slovak National Node of Biobanks
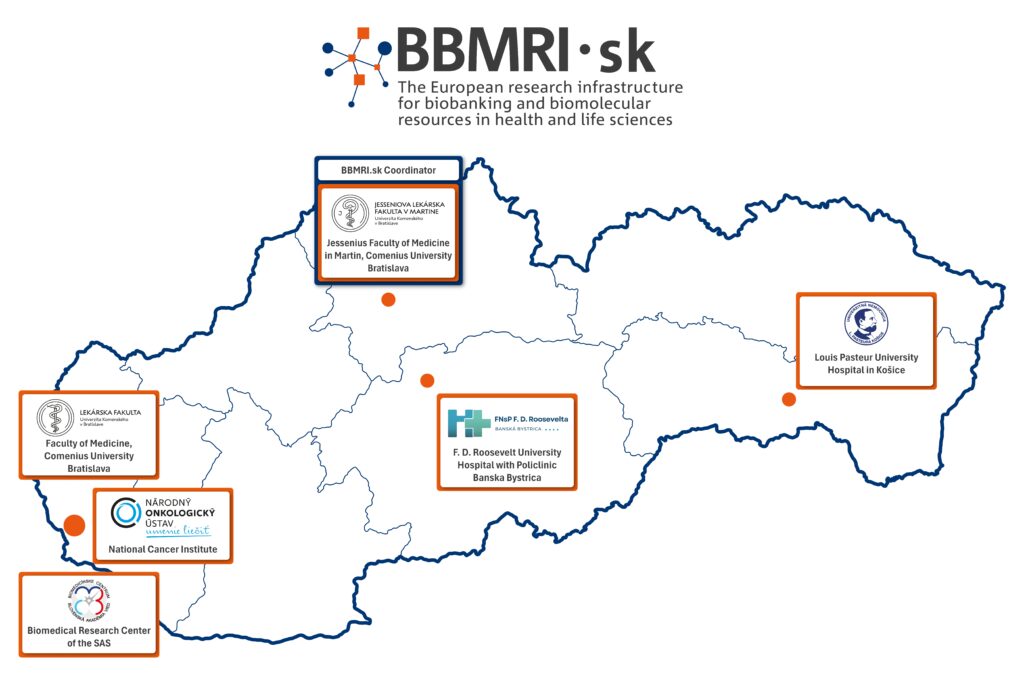
BBMRI.sk is coordinated by the Biobank for Cancer and Rare Diseases, a specialised unit of the Jessenius Faculty of Medicine in Martin, Comenius University Bratislava, which, together with its partners, forms the foundation of the national biobanking network.
BBMRI.sk partners:
Jessenius Faculty of Medicine in Martin, Comenius University Bratislava
Louis Pasteur University Hospital Košice
F. D. Roosevelt University Hospital with Polyclinic Banská Bystrica
Faculty of Medicine, Comenius University Bratislava
National Cancer Institute Bratislava
Biomedical Research Center of the Slovak Academy of Sciences
Together, we are building an infrastructure that enables the ethical, high-quality, and standardised collection and sharing of biological samples for research in Slovakia and across Europe.
Mission
We connect Slovak biobanks and research institutions to support innovative research, improve public health, and ensure the high-quality and ethical use of biological samples and data.
Vision
To build a trusted national biobanking network that contributes to personalized medicine and a healthier society — both in Slovakia and across Europe.
BBMRI-ERIC
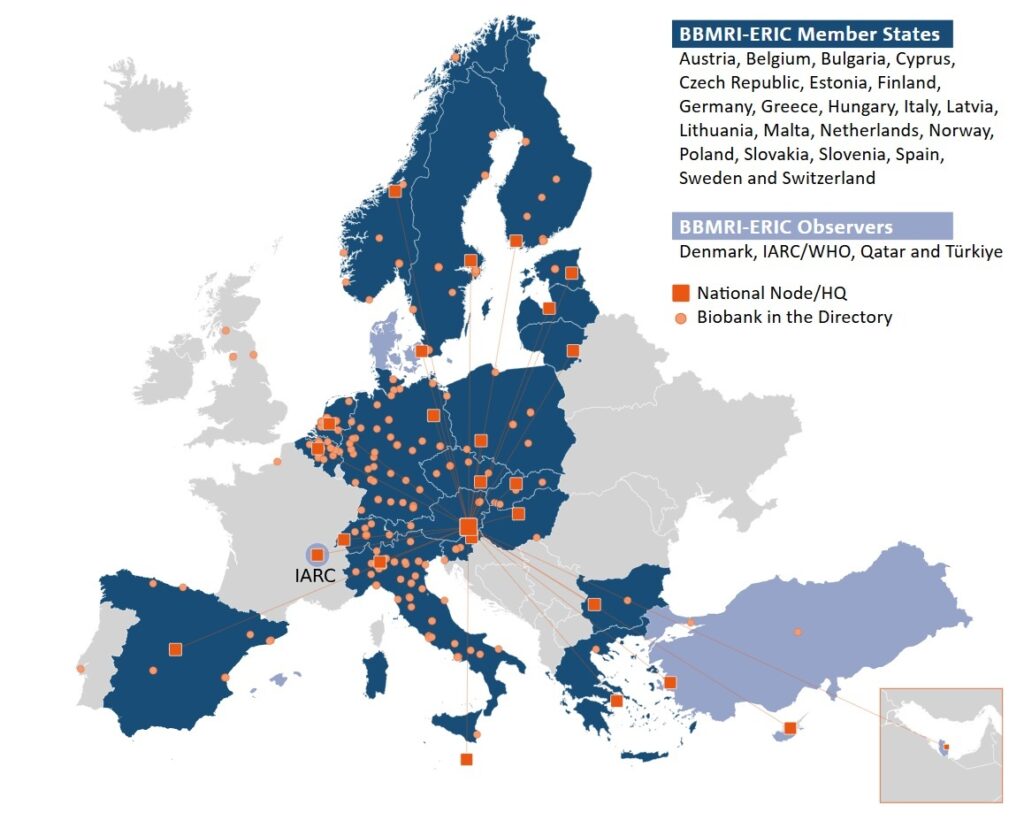
Since 2013, it has served as a connecting platform between biobanks and the European research community. For its 26 members (23 full members and 3 observers), it provides technical tools, expert guidelines, certified standards, and support for cross-border collaboration. Its mission is to facilitate access to high-quality biological samples and related data for scientific and clinical research in accordance with the highest standards. Key activities include the standardised collection, processing, long-term storage, and sharing of biological materials and associated data in compliance with quality requirements, FAIR principles, and the GDPR legislative framework.
Biobanks
Use of Biological Material
Benefits of BBMRI.sk
- Coordinates Slovak biobanks and harmonises their procedures in line with European standards.
- Represents Slovakia within BBMRI-ERIC, ensuring integration with European infrastructure and international projects.
- Assists research teams in identifying suitable samples and data for scientific projects.
- Enhances the quality of biobanking through the implementation of best practices, certifications, and training programmes.
- Creates opportunities for collaboration among academic, clinical, and industrial partners.
- Provides expert know-how in the fields of ethics, legislation, GDPR, data security, and quality management.
- Supports researchers and universities requiring access to reliable biological material.
- Assists healthcare institutions in establishing or developing their own biobanks.
- Helps public institutions develop strategies for science, research, and healthcare.
- Connects the biotechnology and pharmaceutical sectors with partners for innovative projects.
- Ensures transparency and the ethical handling of biological samples for patient organisations and the wider public.
- BBMRI.sk advances Slovak biomedical research to the European level by enabling participation in international projects and clinical studies, thereby opening new opportunities for broader scientific collaboration. At the same time, it improves the quality and availability of research resources, strengthening the capacity of experts to carry out demanding scientific tasks. In doing so, it contributes to the development of personalised and precision medicine, delivering more accurate diagnostics and more effective therapeutic approaches.
- In addition, BBMRI.sk supports innovation and the transfer of knowledge into practice, accelerating the application of the latest scientific findings in clinical settings. Finally, it strengthens public and patient trust in the responsible and secure use of biological samples, which is essential for the sustainable and ethical development of biobanking in Slovakia.
Connection to Europe
To integrate Slovak biobanks into the broader European infrastructure that supports innovation, research, and the development of precision medicine, the Slovak national network:
- shares standards with European partners,
- participates in international research projects,
- provides Slovak institutions with access to European expert groups, certifications, and tools,
- enables researchers to identify samples in foreign biobanks or make Slovak samples available to the European research community.
Organisational Structure
The organisational structure of BBMRI.sk is based on the models of successful national nodes of BBMRI-ERIC across Europe. It is built upon transparent governance, expert working groups, and coordinated services that ensure the harmonised operation of biobanks and effective support for researchers.
Ethics of Sample Use
International Context and Collaboration
Membership of BBMRI.sk in BBMRI-ERIC provides Slovakia with access to harmonised standards, shared tools (such as the BBMRI Directory and Negotiator), and a broad international research community. Participating institutions are therefore able to take part in European projects, make use of expert services in the fields of quality management, ELSI, and data standards, and actively contribute to the advancement of biobanking at the international level.
This membership strengthens Slovakia’s integration into the European Research Area and expands opportunities for collaboration for domestic biobanks and researchers.