Samples
Storage of Biological Samples and Data
Biobanks associated within BBMRI.sk store human biological material together with linked clinical and other related data. Biological samples are catalogued into organized collections that represent a valuable resource for current and future biomedical research. By linking biological samples with relevant data, comprehensive sample/data sets are created, which can be used in research projects both in Slovakia and internationally.
Sample Life Cycle
Within BBMRI.sk, we emphasize that the quality of research is directly dependent on the quality of the biological material used. This quality is influenced by the level of processes carried out throughout the entire sample life cycle:
- Sample indication and collection – biological material is obtained according to established medical procedures.
- Processing and characterization – the sample is professionally processed and complemented with relevant clinical and related data.
- Data continuity – data are securely recorded in hospital/biobank information systems under the supervision of authorized personnel.
- Cataloguing and long-term storage – the material is included in a collection and stored under controlled conditions with systematic quality monitoring.
- Controlled access – biological material is made available exclusively to authorized applicants and only when ethical and methodological requirements are fulfilled, while ensuring the protection of both sample integrity and research data.
This standardized process ensures that samples and their associated data meet high standards of quality, comparability, and usability for modern biomedical research.
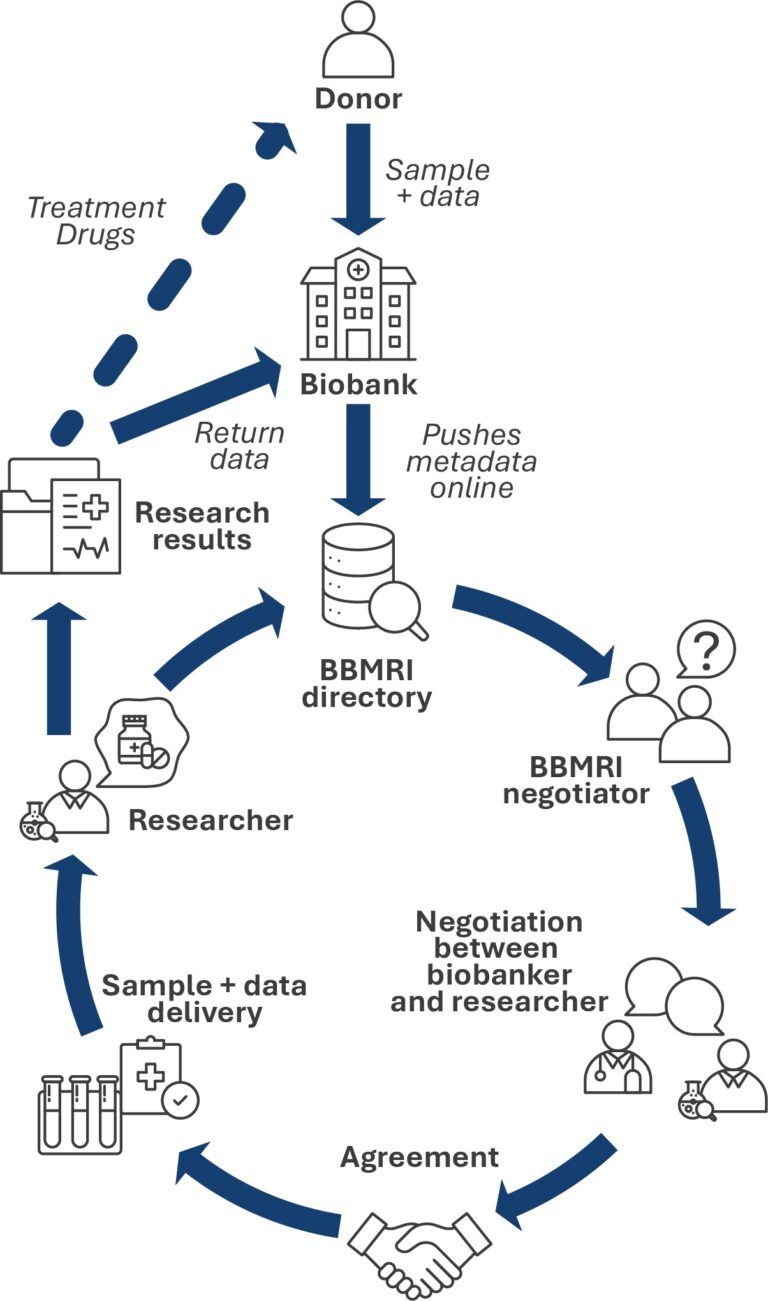
Access to Samples: DIRECTORY, LOCATOR, NEGOTIATOR
The federated Directory platform serves as the largest European catalogue of biobanks, providing an overview of available samples and data. It contains information about biobanks, their collections, sample and data types, as well as conditions of access and quality standards.
Each biobank within the BBMRI-ERIC network shares summary, anonymized metadata about its collections, which are gathered within the central BBMRI-ERIC Directory service.
Instructions on how to search within the Directory are available in the Guide for Working with the Directory.
The BBMRI-ERIC Locator service enables users to search for available biobank resources, samples, or data across Europe within specific biobanks, using existing scientific or institutional accounts – no separate registration is required.
You can sign in using one of the following methods:
LS Login – if you are affiliated with a research or academic institution participating in Life Science infrastructures,
ORCID – using your ORCID ID, commonly used within the scientific community,
other supported identities (e.g., institutional login).
If the Locator identifies samples or collections of interest to a researcher, the communication and access tool Negotiator comes into play. It:
facilitates communication between the researcher and multiple biobanks simultaneously,
is connected to the Directory and automatically imports selected collections,
manages the request process for samples or data,
enables the process to proceed all the way to legal documentation (MTA/DTA).
We recommend reading the Negotiator Guide provided by BBMRI.cz for detailed instructions on using the Negotiator tool.
Types of the samples
Data and samples
QM and SOPs
Biobanks provide a wide range of high-quality biological material for research purposes. Key sample types include blood and its components (serum, plasma, buffy coat), tissues, DNA/RNA, cell lines, and other specialized materials.
The portfolio covers various research areas, including oncology, metabolic diseases, rare diseases, infectious diseases, and population cohorts.
Each sample is linked to relevant data that enable its use in research. Available information may include basic demographic data, clinical diagnosis, medical history, laboratory results, follow-up information, and omics data (genomics, proteomics, metabolomics).
The scope of provided data depends on the specific biobank and the conditions under which the research project is approved.
Biobanks follow strict quality procedures to ensure the reliability of biological samples. Processes are managed through SOPs (Standard Operating Procedures), while biobank personnel perform quality control, process validation, and regular audits.
Many biobanks operate in accordance with the ISO 20387 Biobanking standard, ensuring consistency and reproducibility of processes across the network.
FAIR Access and Use of Samples
Access to samples and associated data is governed by the FAIR principles (Findable, Accessible, Interoperable, Reusable). Biological material may be used only within approved research projects, which require ethical approval and the conclusion of relevant material and data transfer agreements (MTA, DTA).
The subsequent use of biological samples may be restricted depending on the type of study or the conditions of a particular biobank, including possible limitations on commercial use.
All publications and research outputs based on biobank samples must properly acknowledge and cite the respective biobank.
Ethics and Personal Data Protection
Types of the samples
Data and samples
QM and SOPs
Biobanks provide a wide range of high-quality biological material for research purposes. Key sample types include blood and its components (serum, plasma, buffy coat), tissues, DNA/RNA, cell lines, and other specialized materials.
The portfolio covers various research areas, including oncology, metabolic diseases, rare diseases, infectious diseases, and population cohorts.
Each sample is linked to relevant data that enable its use in research. Available information may include basic demographic data, clinical diagnosis, medical history, laboratory results, follow-up information, and omics data (genomics, proteomics, metabolomics).
The scope of provided data depends on the specific biobank and the conditions under which the research project is approved.
Biobanks follow strict quality procedures to ensure the reliability of biological samples. Processes are managed through SOPs (Standard Operating Procedures), while biobank personnel perform quality control, process validation, and regular audits.
Many biobanks operate in accordance with the ISO 20387 Biobanking standard, ensuring consistency and reproducibility of processes across the network.
FAIR Access and Use of Samples
Access to samples and associated data is governed by the FAIR principles (Findable, Accessible, Interoperable, Reusable). Biological material may be used only within approved research projects, which require ethical approval and the conclusion of relevant material and data transfer agreements (MTA, DTA).
The subsequent use of biological samples may be restricted depending on the type of study or the conditions of a particular biobank, including possible limitations on commercial use.
All publications and research outputs based on biobank samples must properly acknowledge and cite the respective biobank.